 Karnataka SSLC Science Answer Key 2026 LIVE
Karnataka SSLC Science Answer Key 2026 LIVE
Karnataka SSLC Science exam is over. It was conducted today, March 23, 2026, from 10 AM to 1:15 PM
across various exam centres. Students are now coming out of their exam halls.
The unofficial answer key is now available here to help students estimate their scores.
As per the reviews collected by multiple test takers, the overall difficulty level of the Science exam has been reported to be
Easy but Lengthy.
Many students felt the 3-hour 15-minute window was just enough, but those who struggled with Physics numericals found themselves rushing toward the end. Subject experts and teachers noted that while the paper was strictly based on the syllabus, it required higher-order thinking skills compared to previous years.
Karnataka SSLC Science Answer Key 2026 (Unofficial)
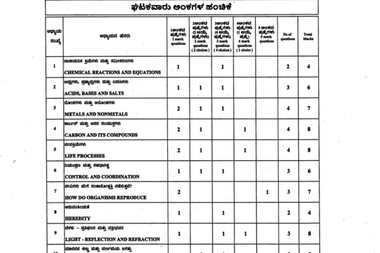
The unofficial Karnataka SSLC Science answer key 2026 is being updated in the table below:
| Question | Answer |
|---|---|
| 1. The power of a lens is +2D. The focal length of this lens is: | (A) 0.5 m |
| 2. The resistivities of four substances (K, L, M, N) at 20°C are given in the table. Which of these is the best conductor? | (C) M |
| 13. The gas liberated when dilute sulphuric acid is added to zinc is: | (B) Hydrogen |
| 14. To make pure iron hard and strong: | (D) carbon will be added in definite proportion |
| 15. The unique property of carbon to form a large molecule by forming bonds with other carbon atoms is: | (C) Catenation |
| 26. The important role of stomata in transportation of water is: | (D) creating upward tension |
| 27. As the food moves forward in the human digestive system the enzymes that help for the digestion respectively are: | (B) Amylase → Pepsin → Lypase |
| 28. Observe the given diagram. The response shown by part X is: | (A) Positive thigmotropism and directional movement |
Stay tuned to this LIVE blog for the Karnataka SSLC Science 2026 unofficial answer key, student reactions, paper analysis, etc!
Karnataka SSLC Science Exam 2026 LIVE
03 00 PM IST - 23 Mar'26
Karnataka SSLC Science Exam Live Coverage Ends
The live coverage for the exam has now concluded. Thank you for following the updates!
02 30 PM IST - 23 Mar'26
Unofficial Answer Key Out
The unofficial answer key for the Karnataka SSLC Science Exam 2026 has been provided in this blog. Students can now check their responses and get an estimate of their expected scores.
02 00 PM IST - 23 Mar'26
Section-Wise Paper Analysis
You can check out the section-wise analysis of Karnataka SSLC Science exam 2026 below:
Section Difficulty Level Physics Moderate Chemistry Easy to Moderate Biology Easy 01 35 PM IST - 23 Mar'26
Unofficial Answer Key & Paper Analysis Coming Up Shortly
Stay tuned as the unofficial answer key and detailed paper analysis will be shared in this LIVE blog shortly. Updates on the overall difficulty level and student reactions will also be provided.
01 15 PM IST - 23 Mar'26
Karnataka SSLC Science Exam 2026 Ends
Karnataka SSLC Science Exam 2026 Ends. Soon the unofficial answer key for the 1-mark questions will be added here.
12 00 PM IST - 23 Mar'26
Karnataka SSLC Science Exam 2026: Result Date
KSEAB will likely release the Karnataka SSLC 2026 result after 35 to 45 days from the last date of examination. This means the result will be released inthe second week of May, 2026.
11 00 AM IST - 23 Mar'26
Karnataka SSLC Science Exam 2026: Upcoming Exam Date
On March 25, 2026, the Karnataka SSLC Second Language exam will take place. The exam will end on April 3, 2026, with the third language exam.
10 00 AM IST - 23 Mar'26
Karnataka SSLC Science Exam 2026: Exam Starts
Students got the Karnataka SSLC Science question papers. till 10.15 AM they can read the questions and post that they can start answering.
09 00 AM IST - 23 Mar'26
Karnataka SSLC Science Exam 2026: Reporting Starts
The reporting process for today's Karnataka SSLC Science Exam 2026 has started. The gate will be closed at 9.45 AM. Post that noone will get permission to enter inside the exam centre.
08 00 AM IST - 23 Mar'26
List of Items Not to Carry
- Electronic Gadgets (Strictly Banned)
- Study Materials & Paper
- Personal Belongings
07 00 AM IST - 23 Mar'26
Short Notes onThe "Nephron"
Candidates should keep in mind the following flow:
Bowman’s Capsule
Glomerulus
Tubular part
06 00 AM IST - 23 Mar'26
Karnataka SSLC Science Exam 2026: Topper-Level Challenge (Biology)
The "Evolution" Evidence (Biology)
Q: Why are the wings of a butterfly and the wings of a bat called "Analogous organs" and not "Homologous"?
05 00 AM IST - 23 Mar'26
Karnataka SSLC Science Exam 2026: Topper-Level Challenge (Chemistry)
The "Amphoteric" Puzzle (Chemistry)
Q: You are given three metal oxides: NA2O, Al2O3, and SO2. Which one will react with both HCl and NaOH? Write the chemical equations.
04 00 AM IST - 23 Mar'26
Karnataka SSLC Science Exam 2026: Topper-Level Challenge (Physics)
The "Copper Wire" Logic (Physics)
Q: A wire of resistance R is cut into five equal parts. These parts are then connected in parallel. If the equivalent resistance of this combination is R', what is the ratio R/R2?
The "Light" Ray Trap (Physics)
Q: A ray of light falls normally (at 90°) on the surface of a rectangular glass slab. What is the angle of incidence and the angle of refraction?
03 00 AM IST - 23 Mar'26
High-Priority "Must-Watch" Topics for 2026
- Electricity
- Acids & Bases
- Heredity
- Magnetic Effects
- Reproduction
02 00 AM IST - 23 Mar'26
The "Last 15 Minutes" Topper Routine
- Unit Sweep: Make sure you wrote "Dioptre" for power and "Ohm" for resistance.
- Question Numbering: It is mandatory to correctly write the sub-question numbers, and the same should be matched with the question paper exactly.
- The "Pencil Line": Draw a horizontal line with a pencil after every completed answer.
01 00 AM IST - 23 Mar'26
Toppers Tips Regarding Chemistry: The "Equation" Strategy
- Always balance the equation
- Explain the symbols
- In Carbon compounds, do not forget to show the bonds
12 00 AM IST - 23 Mar'26
Sample Practice Questions on Light-Reflection and Refraction (1/3)
Ques. If the length of a wire is doubled and its area of cross-section is halved, its resistance will:
Answer. Increase four times
Ques. An ammeter is always connected in ________ in a circuit, while a voltmeter is connected in ________.
Answer. Series, Parallel
Ques. The rate at which electric energy is dissipated or consumed in an electric circuit is called:
Answer. Electric Power
11 00 PM IST - 22 Mar'26
Sample Practice Questions on Light-Reflection and Refraction (1/2)
Ques. Calculate the electrical energy consumed by a 100W bulb used for 10 hours a day for one day.
Answer. 1kWh
Ques. What is the function of a galvanometer in a circuit?
Answer. To detect the presence of current.
Ques. Which physical quantity has the unit 'Watt-second'?
Answer. Electric Energy
10 00 PM IST - 22 Mar'26
Sample Practice Questions on Light-Reflection and Refraction (1/1)
Ques. According to Ohm's Law, if the potential difference across a conductor is doubled while keeping the resistance constant, what happens to the current?
Answer. It is doubled.
Ques. Three resistors of 2Ω, 3Ω, and 5Ω are connected in series. What is the equivalent resistance of the circuit?
Answer. 10 Ω
Ques. Why is tungsten used almost exclusively for the filament of electric lamps?
Answer. It has a very high melting point
09 40 PM IST - 22 Mar'26
Sample Practice Questions on Light-Reflection and Refraction (1/3)
Ques. The SI unit of the refractive index is:
Answer. It has no unit
Ques. For a convex mirror, if an object is placed at infinity, the image is formed at:
Answer. The Focus (F) behind the mirror
09 20 PM IST - 22 Mar'26
Sample Practice Questions on Light-Reflection and Refraction (1/2)
Ques. A doctor has prescribed a corrective lens of power +1.5 D. The focal length and type of the lens are:
Answer. +.67 m, Convex Lens
Ques. According to the Cartesian sign convention, the object distance ($u$) is always taken as:
Answer. Negative
Ques. A student uses a lens that produces a virtual, erect, and diminished image regardless of the object's position. This lens is a:
Answer. Concave Lens
09 00 PM IST - 22 Mar'26
Sample Practice Questions on Light-Reflection and Refraction (1/1)
Ques. An object is placed at the center of curvature (C) of a concave mirror. The nature and size of the image formed will be:
Answer. Real, Inverted, and Same size
Ques. A ray of light enters from a rarer medium to a denser medium. How does it bend?
Answer. It bends towards the normal.
Ques. If the magnification produced by a lens is m = -2, what does this indicate about the image?
Answer. The image is real, inverted, and enlarged
08 40 PM IST - 22 Mar'26
Saturated and Unsaturated Carbon Compounds
Alkanes (Saturated): Single bonds only. Formula:
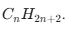
Alkenes (Unsaturated): At least one double bond. Formula:
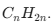
Alkynes (Unsaturated): At least one triple bond. Formula:
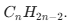
08 20 PM IST - 22 Mar'26
Expert Exam Tip on Human Eye And Colourful World
If the power is Negative (-), the person has Myopia (uses a concave lens).
If the power is Positive (+), the person has Hypermetropia (uses a convex lens).
08 00 PM IST - 22 Mar'26
Defects of Vision (The "Guarantee" Question)
Defect Correction Myopia (Near-sightedness) Concave Lens Hypermetropia (Far-sightedness) Convex Lens Presbyopia Bifocal Lens 07 40 PM IST - 22 Mar'26
The "Final Lap" Strategy for Karnataka SSLC Science Exam 2026
- 8 PM to 10 PM: Practice drawing the 7 Big Biology Diagrams (Heart, Brain, Nephron, Reflex Arc, Flower, Male/Female Reproductive systems).
- 5 AM to 7 AM: Review Chemical Equations (Types of reactions and the Salts table).
07 20 PM IST - 22 Mar'26
What are the sources of Energy?
- Good Source of Energy
- Fossil Fuels
- Biogas (Gobar Gas)
- Solar Energy
- Nuclear Energy
07 00 PM IST - 22 Mar'26
Bonus: The Simple Domestic Circuit
Live Wire (Red): 220V potential.
Neutral Wire (Black): Zero potential.
Earth Wire (Green): Connected to a metal plate deep in the earth for safety.
Parallel Connection: All appliances are connected in parallel; hence, they get the same voltage and work independently.
06 40 PM IST - 22 Mar'26
Must-Label Parts for Full Marks
Armature Coil (ABCD): The rectangular coil of insulated copper wire.
Strong Magnets (N & S): To provide a permanent magnetic field.
Split Rings / Commutator (P & Q): Two halves of a metallic ring.
Brushes (X & Y): Carbon blocks that touch the rings to provide current.
Battery & Switch: To complete the circuit.
06 20 PM IST - 22 Mar'26
The Three "Hand Rules" on Magnetic Effects Of Electric Current
- The right-hand thumb rule helps to find the direction of the Magnetic Field around a straight current-carrying conductor.
- Fleming’s Left-Hand Rule (Motor Rule) is used to find the direction of Force/Motion.
- Fleming’s Right-Hand Rule (Generator Rule) is used to find the direction of the induced current.
06 00 PM IST - 22 Mar'26
Final Study Tip about "Chlor-Alkali"
Hydrogen (at Cathode)
Chlorine (at Anode)
Sodium Hydroxide (near Cathode)
05 40 PM IST - 22 Mar'26
Key Chemical Reactions to Remember
Chlor-Alkali Process 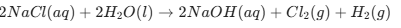
Making Bleaching Powder 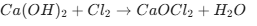
Heating Baking Soda 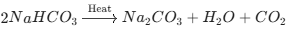
Plaster of Paris to Gypsum 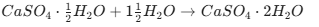
05 20 PM IST - 22 Mar'26
Important Salts (The "High-Score" Table)
Common Name Chemical Name Formula Common Salt Sodium Chloride NaCl Bleaching Powder Calcium Oxychloride CaOCl2 Baking Soda Sodium Hydrogen Carbonate NaHCO3 Washing Soda Sodium Carbonate Decahydrate Na2CO3.10H2O Plaster of Paris Calcium Sulphate Hemihydrate CASO4.1/2H2O Gypsum Calcium Sulphate Dihydrate CaSO4.2H2O 05 00 PM IST - 22 Mar'26
List of Common Acids & Bases
Common Name Chemical Name Formula Hydrochloric Acid Hydrogen Chloride HCl Sulphuric Acid Hydrogen Sulphate H2SO4 Nitric Acid Hydrogen Nitrate HNO3 Acetic Acid Ethanoic Acid CH3COOH Caustic Soda Sodium Hydroxide NaOH Milk of Magnesia Magnesium Hydroxide Mg(OH)2 Slaked Lime Calcium Hydroxide Ca(OH)2 04 45 PM IST - 22 Mar'26
Final "Acids & Bases" Checklist for Monday:
- Antacids: Magnesium Hydroxide is a weak base, which is used to neutralize stomach acid.
- Universal Indicator: A mixture of indicators that shows different colors at different pH values.
- Neutralization: Acid + Base ---------> Salt + Water.
04 30 PM IST - 22 Mar'26
Dilution: The Safety Rule
The board usually asks questions like "Why should water be added to acid, or acid to water?"
The Rule: Always add Acid to Water drop-by-drop with constant stirring.
04 15 PM IST - 22 Mar'26
The pH Calculation (A Hidden Numerical)
Question: Solution A has a pH of 2, and Solution B has a pH of 6. Which one has a higher concentration of H+ ions?
Logic: The lower the pH, the higher the H+ concentration.
04 00 PM IST - 22 Mar'26
Strong vs. Weak Acids/Bases
Particulars Strong Acid Weak Acid Acid Type Completely dissociates (breaks apart) in water Only partially dissociates in water. Example HCl, H2SO4, HNO3 CH3COOH (Acetic acid/Vinegar), Citric acid. Observation High concentration of H+ ions. Low concentration of H+ ions. 03 45 PM IST - 22 Mar'26
Indicators & Color Changes from Acids, Bases and Salts
Indicator Acid Color Base Color Litmus Red Blue Methyl Orange Red Yellow Phenolphthalein Colorless Pink Turmeric Yellow Reddish-brown 03 30 PM IST - 22 Mar'26
The "Big Four" Salts (4-Mark Potential)
Salt Name Chemical Formula Key Use Bleaching Powder CaOCl2 Disinfecting water; bleaching cotton. Baking Soda NaHCO3 An ingredient in antacids. Washing Soda Na2CO3. 10H2O Remove the permanent hardness of water Plaster of Paris CaSO4.1/2H2O Supports the fractured bones; use in making toys 03 15 PM IST - 22 Mar'26
List of Practice Questions on How Do Organisms Reproduce? (1/2)
Ques. What happens if the egg is not fertilized in a human female?
Answer. Menstruation occurs
Ques. In the context of DNA copying during reproduction, what is the significance of 'variation'?
Answer. It helps in the survival of species over time.
Ques. Which contraceptive method involves the surgical blocking of the vas deferens?
Answer. Vasectomy
Ques. Vegetative propagation refers to the formation of new plants from:
Answer. Stems, roots, and leaves
03 00 PM IST - 22 Mar'26
List of Practice Questions on How Do Organisms Reproduce? (1/1)
Ques.In flowering plants, which part eventually develops into the seed after fertilization?
Answer. Ovule
Ques. Why are the testes located in the scrotum outside the abdominal cavity in human males?
Answer. To maintain a temperature lower than the body temperature.
Ques. Which method of asexual reproduction is seen in Rhizopus (bread mould)?
Answer. Spore formation
Ques. The placenta is a specialized tissue in human females that is responsible for:
Answer. Providing nutrition and oxygen to the embryo.
02 45 PM IST - 22 Mar'26
The "Exam Morning" Checklist
- Check whether the candidates take all the required stationery items, such as 2-sharpened HB pencils, an eraser, and a blue/black ballpoint pen.
- Write the Values given, draw the Jagged (Rough) diagram if required, and write the Definition/Formula before solving.
- Do not overwrite a mistake. Instead, draw a single neat line through it. Do not scribble.
- Carry your own transparent water bottle.
02 30 PM IST - 22 Mar'26
Your "Sunday Night" Routine
- Do not read any new topic.
- Spend 10 minutes recalling the formula flash.
- Can you draw the Heart and Nephron in under 3 minutes? If yes, then you are ready for the exam.
- Your brain converts "short-term" study into "long-term" memory while you sleep. Hence, the candidates should sleep for 7 to 8 hours.
02 15 PM IST - 22 Mar'26
The "Motor" vs. "Generator" Logic
Particulars Details Electric Motor Converts Electrical -------> Mechanical energy Electric Generator Converts Mechanical ---------> Electrical energy 02 00 PM IST - 22 Mar'26
Final Words of Encouragement
Sections Final Words Physics Numericals, Signs, and Laws. Biology Life processes, Diagrams, and Mendel’s genetics. Chemistry Reactions, Carbon structures, and Metallurgy. 01 45 PM IST - 22 Mar'26
"Final 5" Rapid-Fire Check
Particulars Details The "Mirror" vs. "Lens" Sign Trap Question: If a magnification is 2, what does it tell you? Magnetic Field Lines Question: Why do two magnetic field lines never intersect? The "Alkali" Distinction Question: Is every base an alkali? Resistance in Parallel vs. Series Question: Why is parallel transition preferred in domestic (home) circuits? Biodegradable vs. Non-Biodegradable Question: Why can't enzymes break down plastics? 01 30 PM IST - 22 Mar'26
Karnataka SSLC Science Exam 2026: Final Expert Checklist
- The "15-Minute Rule": Spend 15 minutes reading the questions carefully. Tick the numericals which they are 100% sure of.
- Order of Writing: Start the section, which is the strongest section of the candidates.
- Diagrams First: If the long answer involves a diagram, then the candidates should draw it first. It will also help the candidates in mental mapping, so that they can write the answers properly.
01 15 PM IST - 22 Mar'26
Chemical Properties: The Reaction Zone
- Reaction with Oxygen
- Reaction with Water
- Reactivity Series
01 00 PM IST - 22 Mar'26
Short Notes on Animal Hormones (Endocrine System) (1/2)
Gland Hormone Function Pancreas Insulin Regulates blood sugar levels. Adrenal Adrenaline Increases heartbeat. Testes/Ovaries Testosterone/Estrogen Development of secondary sexual characteristics. 12 45 PM IST - 22 Mar'26
Short Notes on Animal Hormones (Endocrine System) (1/1)
Gland Hormone Function Pituitary Growth Hormone Regulates the growth and development Thyroid Thyroxine Regulates metabolism (requires Iodine). 12 30 PM IST - 22 Mar'26
Short Notes on Plant Hormones (Phytohormones)
Hormone Primary Function Auxin Helps in cell elongation and is responsible for phototropism. Gibberellin Helps in stem growth Cytokinin Promotes cell division Abscisic Acid Inhibits growth; causes wilting of leaves 12 15 PM IST - 22 Mar'26
Important Chemical Properties (The 3-Mark Zone) on Carbon And Its Compounds
Combustion: Carbon compounds burn in oxygen to produce CO2, H20, heat, and light.
Addition Reaction: Conversion of unsaturated fats into saturated fats using a Nickel catalyst.
Substitution Reaction: Saturated hydrocarbons react with chlorine under sunlight.
Esterification: Reaction between Ethanoic acid and Ethanol to form a sweet-smelling Ester.
12 00 PM IST - 22 Mar'26
Functional Groups & Nomenclature: Carbon and Its Compounds
- Alcohol: - OH — e.g., Ethanol.
- Aldehyde: - CHO — e.g., Methanal.
- Ketone: >C=O — e.g., Propanone.
- Carboxylic Acid: -COOH — e.g., Ethanoic acid.
11 45 AM IST - 22 Mar'26
Short Notes on The "Versatile Nature" of Carbon (2-3 Marks)
This is one of the high probability questions, and the candidates should explain the reasons why carbon forms so many compounds. Here are the reasons:
- Catenation
- Tetravalency
11 30 AM IST - 22 Mar'26
Experts' guidance on Carbon and Its Compounds
- While you draw any structure, do not forget to count the bonds for every carbon atom
- It must always have exactly four lines (bonds) connected to it.
- Nonetheless, if you draw five or three lines, then you will lose the entire marks for drawing the incorrect structures
11 15 AM IST - 22 Mar'26
Final Strategy for Biology
- If there is a question on a process, then the candidates should add a small and neat sketch (even if it is not mentioned in the question paper). It helps you to get full marks.
- Keywords: Use terms like Peristalsis, Alveoli, Villi, and Synapse.
11 00 AM IST - 22 Mar'26
Top 5 Biological Processes for Karnataka SSLC Science Exam 2026
- Double Circulation in the Human Heart
- Excretion and the Function of Nephrons
- Reflex Action and the Reflex Arc
- Aerobic vs. Anaerobic Respiration
- Transport in Plants: Xylem vs. Phloem
10 45 AM IST - 22 Mar'26
Expert "Last-Minute" Refraction Check
- Case 1 (Rarer to Denser): Light moving from Air to Glass bends towards the normal (∠i > ∠r).
- Case 2 (Denser to Rarer): Light moving from Glass to Air bends away from the normal (∠i < ∠r).
- Snell's Law: Always remember sin i/ sin r = constant (Refractive Index).
10 30 AM IST - 22 Mar'26
Sample Question on Refractive Index (The Speed of Light)
Question: The refractive index of glass is 1.50, and the speed of light in air is 3X108 m/s. Calculate the speed of light in glass.
Final Conclusion: The speed of light in glass is 2X108 m/s
10 15 AM IST - 22 Mar'26
Sample Question on Power of a Lens
Question: A doctor has prescribed a corrective lens of power +1.5D. Find the focal length of the lens. Is the prescribed lens diverging or converging?
Answer.
Focal length: +66.6 cm.
Nature: Since the power and focal length are positive, it is a Converging Lens (Convex Lens).
10 00 AM IST - 22 Mar'26
Common Conceptual Questions on Light-Reflection and Refraction
- Why is a convex mirror used as a rear-view mirror?
- Define the Principal Focus of a concave mirror.
- Why does a pencil appear bent when dipped in water?
09 45 AM IST - 22 Mar'26
Formula Bank & Numerical Tips of Light-Reflection and Refraction
Concept Mirror Formula Lens Formula Formula 1/f= 1/v+1/u 1/f= 1/v-1/u Magnification (m) m = - v/u m = v/u Height Ratio m= h'/h m= h'/h 09 30 AM IST - 22 Mar'26
Karnataka SSLC Science: Chemical Reactions Quiz (1/2)
Ques. What type of reaction occurs when Quicklime ($CaO$) reacts with water?
Answer. Exothermic Combination
Ques. What is the observation when an iron nail is placed in a Copper Sulphate solution?
Answer. The blue solution turns pale green, and a brown coating forms on the nail.
09 15 AM IST - 22 Mar'26
Karnataka SSLC Science: Chemical Reactions Quiz (1/1)
Ques. When Ferrous Sulphate crystals are heated in a dry boiling tube, what is the color change observed in the residue?
Answer. Green to Reddish-brown
Ques. Which gas is evolved as 'brown fumes' during the thermal decomposition of Lead Nitrate?
Answer. Nitrogen Dioxide
Ques. In the electrolysis of water, why is the volume of gas collected at one electrode double the volume collected at the other?
Answer. Water contains two parts of Hydrogen for every one part of Oxygen
09 00 AM IST - 22 Mar'26
Final 10 List of the Most Important Chemical Reactions (1/3)
Topic Name Chemical Reactions Reaction of Ethanol with Sodium 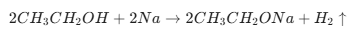
Esterification Reaction 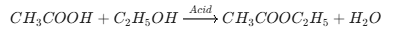
Saponification (Soap formation) 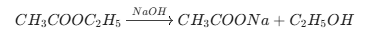
08 45 AM IST - 22 Mar'26
Final 10 List of the Most Important Chemical Reactions (1/2)
Topic Name Chemical Reactions Iron Nail in Copper Sulphate Solution 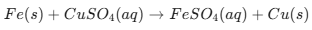
Sodium Sulphate and Barium Chloride 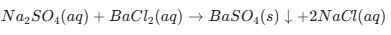
Thermite Reaction 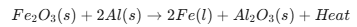
08 30 AM IST - 22 Mar'26
Final 10 List of the Most Important Chemical Reactions (1/1)
Topic Name Chemical Reactions Decomposition of Ferrous Sulphate (Thermal) 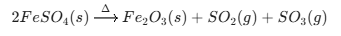
Decomposition of Lead Nitrate 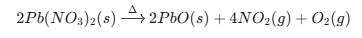
Electrolysis of Water 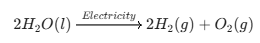
Reaction of Quicklime with Water 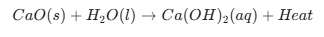
08 15 AM IST - 22 Mar'26
Three Scoring Areas of The Human Eye & Colourful World
- Defects of Vision
- Myopia (Near-sightedness)
- Hypermetropia (Far-sightedness)
- Dispersion & Scattering
- Dispersion
- Tyndall Effect
- Atmospheric Refraction
- Defects of Vision
08 00 AM IST - 22 Mar'26
Karnataka SSLC Science Exam 2026: Time Management Strategy
Particulars Time Allotted Reading Time 15 minutes MCQs & 1-Mark 30 minutes 2-Mark Questions 40 minutes 3-Mark Questions 50 minutes 4 & 5-Mark Questions 45 minutes Revision 13 minutes 07 45 AM IST - 22 Mar'26
The "Golden Rules" of Sign Convention
Object Distance (u): Always Negative (-).
Focal Length (f):
Concave (Mirror/Lens) = Negative (-)
Convex (Mirror/Lens) = Positive (+)
Real Image: v is negative for mirrors, positive for lenses.
Virtual Image: v is positive for mirrors, negative for lenses.
07 30 AM IST - 22 Mar'26
Total Resistance and the Total Current: Problem 1
The Problem:
In the given circuit, three resistors R1= 10Ω, R2= 20Ω, and R3= 60Ω are connected in parallel to a 24V battery.
- Calculate the Total Resistance (Rp) of the circuit.
- Calculate the Total Current (I) flowing through the circuit.
07 15 AM IST - 22 Mar'26
Common "Pitfalls" to Avoid
- Time Conversion: In H= I2Rt or P= W/t, time must be written in seconds.
- Ammeter/Voltmeter Placement: Do not swap these two. An ammeter is always in series; a voltmeter is always in parallel.
- Parallel Calculation: When solving 1/Rρ= 1/2+1/2 = 1, then students often forget the final step is to find Rρ. If 1/Rρ= 1/4, then Rρ= 4Ω.
07 00 AM IST - 22 Mar'26
Frequently Asked "Application" Questions
- Why is Tungsten used for filaments?
- Why does the resistance of a conductor increase with length?
- Why are domestic circuits connected in parallel?
06 45 AM IST - 22 Mar'26
Must-Know Formulas on Electricity (1/2)
Concept Formula Key Units Joule's Heating H= I2Rt H (Joules), t (Seconds) Electric Power P=VI or P=I2R P (Watts) 06 30 AM IST - 22 Mar'26
Must-Know Formulas on Electricity (1/1)
Concept Formula Key Units Ohm's Law V=IR V (Volts), I (Amperes), R (Ohms) Resistivity R= ρl/A ρ (Ohm-meter), l (m), A (m2) Series Resistance RS= R1+R2+R3 Total resistance increases Parallel Resistance 1/Rρ= 1/R1+1/R2+1/R3 Total resistance decreases 06 15 AM IST - 22 Mar'26
Experts' Guidance on Electricity
Expert Strategy: The Three Pillars
- Zone 1: Definitions & SI Units (for 1-2 Marks questions)
- Zone 2: Diagrams & Graphs (for 2-3 Marks questions)
- Zone 3: High-Value Numericals (for 3-4 Marks questions)
06 00 AM IST - 22 Mar'26
Last-Minute Strategy for Diagrams
- Use a sharp pencil. Try to avoid a pen for drawing the diagrams.
- Try to add labels on the right side of the diagram using a straight line to create a cleaner look.
- Note in Physics ray diagrams, an omitted arrowhead (which shows the light direction) may cost a half mark.
05 45 AM IST - 22 Mar'26
Most Frequently Asked Diagrams from Chemistry
- Electrolysis of Water
- Reaction of Zinc granules with Dilute Sulphuric Acid
- Action of Steam on Metals
05 30 AM IST - 22 Mar'26
Most Frequently Asked Diagrams from Physics
- Ray Diagrams for Concave Mirrors
- Ray Diagrams for Convex Lenses
- The Human Eye
- Electric Motor & Generator
05 15 AM IST - 22 Mar'26
Most Frequently Asked Diagrams from Biology Section
- The Human Digestive System
- The Human Heart
- The Excretory System (Human Urinary System)
- The Human Brain
- Structure of a Flower
05 00 AM IST - 22 Mar'26
Karnataka SSLC Science Exam 2026: Key Preparation Tips
- Diagrams (Biology/Physics): Practice the Human Heart, Brain, Excretory System, and Ray Diagrams for mirrors and lenses.
- Balanced Equations: In Chemistry, candidates should ensure that chemical equations are balanced and include state symbols (s, l, g, aq), where necessary.
- Numericals: Practice the light numericals and electricity questions. Do not forget to mention the SI units in the final answer,otherwise, the candidates might lose 0.5 to 1 mark.













