 TG Inter 2nd Year Chemistry Answer Key 2026 LIVE Updates: Student Reviews; Expert Solved Question Paper
TG Inter 2nd Year Chemistry Answer Key 2026 LIVE Updates: Student Reviews; Expert Solved Question PaperTG Inter 2nd Year Chemistry exam 2026 was held on March 13, 2026 . The exam took place during the morning session from 9 AM to 12 PM . Students arrived at their designated exam centres around 8 AM to 8:30 AM. Hall tickets were already distributed by the respective junior colleges, and students had to carry their printed admit cards and a valid school ID to enter the exam hall. The TG Inter 2nd Year Chemistry Answer Key 2026 has been shared below.
TG Inter 2nd Year Chemistry Answer Key 2026 (Unofficial)
Here is the TG Inter 2nd Year Chemistry Answer Key 2026:Q.No | Question | Answer |
|---|---|---|
1 | State Henry's law. | At a constant temperature, the solubility of a gas in a liquid is directly proportional to the partial pressure of the gas present above the surface of liquid or solution. Formula: P=K_H⋅χ. |
2 | What is a primary battery? Give one example. | A battery in which the redox reaction occurs only once and the battery becomes dead after some time (cannot be recharged). Example: Dry cell or Mercury cell. |
3 | What is the role of cryolite in the metallurgy of aluminium? |
1. It lowers the melting point of the alumina (Al_2O_3) from 2323 K to about 1140 K.
|
4 | Nitrogen molecule is highly stable. Why? | Because it contains a strong triple bond (N≡N) between two nitrogen atoms, which results in a very high bond dissociation enthalpy (941.4 kJ/mol). |
5 | Why Zn2+ is diamagnetic whereas Mn2+ is paramagnetic? | Zn^2+ has a completely filled d-orbital (3d^10) with no unpaired electrons. Mn^2+ has a half-filled d-orbital (3d^5) with 5 unpaired electrons, which causes paramagnetism. |
6 | What is Ziegler-Natta catalyst? | It is a mixture of Titanium tetrachloride (TiCl_4) and Triethylaluminium [Al(C_2H_5)_3]. It is used for the polymerization of ethene into high-density polythene. |
7 | What is PHBV? How is it useful to man? | It is Poly β-hydroxybutyrate-co-β-hydroxyvalerate. It is a biodegradable polymer used in specialty packaging, orthopedic devices, and controlled release of drugs. |
8 | What are antacids? Give example. | Chemical substances which neutralize excess acid in the stomach and raise the pH to an appropriate level. Example: Magnesium hydroxide [Mg(OH)_2] or Omeprazole. |
9 | What are artificial sweetening agents? Give one example. | Chemicals that provide sweetness to food but do not add calories to the body. They are excreted in urine unchanged. Example: Aspartame or Saccharin. |
Section B & Section C Solved in the live coverage below! | ||
TG Inter 2nd Year Chemistry Exam 2026 Quick Facts
The question paper will be divided into three sections. You can check all details and facts related to TG Inter 2nd Year Chemistry 2026 below:
Aspect | Details |
|---|---|
Exam Date | March 13, 2026 |
Exam Time | 9 AM to 12 PM |
Sections | Sections A, B, & C |
Questions and Marking Scheme |
|
Max Marks | 60 Marks |
In this live blog, students can check answer key for TG Inter 2nd Year Chemistry along with analysis and student reactions. Stay tuned for continuous updates!
TG Inter 2nd Year Chemistry Exam 2026 LIVE
01 30 PM IST - 13 Mar'26
Section B, Q 18: (a) What are ambident nucleophiles? (b) What are Enantiomers?
Correct Answer:
(a) Ambident Nucleophiles: Nucleophiles that possess two nucleophilic centers through which they can attack.
Example: Cyanide group CN^- can attack through Carbon to form nitriles or through Nitrogen to form isocyanides.
(b) Enantiomers: Stereoisomers that are non-superimposable mirror images of each other. They rotate plane-polarized light in opposite directions but have identical physical properties like melting and boiling points.
01 25 PM IST - 13 Mar'26
Section B, Q 17: What are hormones? Give one example for each of the following:
Hormones are chemical messengers produced by endocrine glands that travel through the bloodstream to regulate physiological activities.
(a) Steroid hormones: Testosterone or Estrogen.
(b) Polypeptide hormones: Insulin or Glucagon.
(c) Amino acid derivatives: Epinephrine (Adrenaline).
01 20 PM IST - 13 Mar'26
Section B, Q 16: Explain Werner's theory of coordination compounds with suitable examples.
Alfred Werner proposed two types of valencies for metals in coordination compounds:
Primary Valency: Ionizable, corresponds to the oxidation state, and is satisfied by negative ions.
Secondary Valency: Non-ionizable, corresponds to the coordination number, and is satisfied by neutral molecules or negative ions (ligands). It is directional in space.
Example: In [Co(NH)_3)_6]Cl_3, the 3 Cl^- ions represent primary valency, while the 6 NH_3 molecules represent secondary valency.
01 15 PM IST - 13 Mar'26
Section B, Q15: Explain the structures of (a) XeF_2 and (b) XeF_4.
Correct Answer:
(a) XeF_2: Xenon has 8 valence electrons. It forms 2 bond pairs and has 3 lone pairs. Hybridization is sp^3d. The geometry is trigonal bipyramidal, but the actual shape is Linear to minimize lone pair-lone pair repulsions.
(b) XeF_4: Xenon forms 4 bond pairs and has 2 lone pairs. Hybridization is sp^3d^2. The geometry is octahedral, but the shape is Square Planar with lone pairs positioned opposite to each other.
01 10 PM IST - 13 Mar'26
Section B, Q 14: Explain the purification of sulphide ore by froth floatation method.
This method is based on the difference in the wetting characteristics of the ore and gangue particles.
The powdered ore is mixed with water and "collectors" (e.g., pine oil) which enhance the non-wettability of mineral particles.
"Froth stabilizers" (e.g., cresols) are added to sustain the froth.
Air is blown through the mixture, creating froth.
The mineral particles, being wetted by oil, rise to the surface with the froth, while gangue particles sink in water.
01 00 PM IST - 13 Mar'26
Section B, Q11: Classify each of the following as either a p-type or an n-type semiconductor.
Correct Answer:
- (a) Ge doped with In: Germanium (Group 14) doped with Indium (Group 13). Since Indium has only 3 valence electrons, it creates a "hole" or electron vacancy. This is a p-type semiconductor.
(b) Si doped with B: Silicon (Group 14) doped with Boron (Group 13). Similar to the above, Boron creates electron-deficient holes. This is a p-type semiconductor.
12 50 PM IST - 13 Mar'26
Answer Key Released
The answer key of TG Inter 2nd Year Chemistry Exam 2026 has been released! Check the solved questions and verify with your responses to get an idea about the exam performance.
12 00 PM IST - 13 Mar'26
TG Inter 2nd Year Chemistry Exam 2026 Ending Now
The three-hour Chemistry exam is about to conclude shortly. Students will soon begin exiting their exam centres. Detailed question paper analysis, answer key, and student reactions will be shared soon in this live blog. Stay tuned for further updates.
11 00 AM IST - 13 Mar'26
Final Hour of the Chemistry Exam
Students are now in the last hour of the exam. This is the ideal time to finish pending answers and quickly revise the completed sections of the paper.
10 00 AM IST - 13 Mar'26
Exam Currently Underway
The TG Inter 2nd Year Chemistry exam is now in progress across various exam centres in Telangana.
09 00 AM IST - 13 Mar'26
Chemistry Exam Set to Begin Shortly
The TG Inter 2nd Year Chemistry exam will start soon. Students are entering their respective exam halls and settling into their seats. Once the question paper is distributed, read all instructions carefully before attempting the answers.
08 00 AM IST - 13 Mar'26
Students Reaching Their Exam Centres
Students are gradually arriving at their designated exam centres for the TG Inter 2nd Year Chemistry exam. Entry verification and hall ticket checking are expected to begin soon.
07 00 AM IST - 13 Mar'26
Students Starting to Leave for Exam Centres
Across Telangana, many students have begun leaving their homes to reach their allotted exam centres for the Chemistry exam. Before heading out, make sure you carry your hall ticket, ID card, and necessary stationery.
06 00 AM IST - 13 Mar'26
Good Morning Students - Exam Day Has Arrived
Good morning to all students appearing for the TG Inter 2nd Year Chemistry exam today. Stay calm, confident, and positive. Eat a light and healthy breakfast, double-check your hall ticket, and plan to reach the exam centre well before time.
05 00 AM IST - 13 Mar'26
Items Not Allowed at Exam Centres
Students should ensure that they do not carry restricted items to the examination centre, as these may cause issues during checking or entry.
- Mobile phones
- Smart watches or digital watches
- Calculators
- Earphones or Bluetooth devices
- Written notes or textbooks
- Paper chits or loose sheets
- Bags or backpacks inside the exam hall
- Any electronic gadgets
04 00 AM IST - 13 Mar'26
Solved 8-Mark Question for Chemistry
How is chlorine prepared in the laboratory? Explain its reactions with:
(a) Iron
(b) Hot, concentrated NaOH
(c) Na₂S₂O₃Laboratory Preparation of Chlorine
In the laboratory, chlorine gas is prepared by heating manganese dioxide (MnO₂) with concentrated hydrochloric acid (HCl). Since chlorine is heavier than air, it is collected by downward displacement of air.
Reaction:
MnO₂ + 4HCl → MnCl₂ + 2H₂O + Cl₂Reactions of Chlorine
(a) With Iron
Iron reacts with chlorine to form ferric chloride.
2Fe + 3Cl₂ → 2FeCl₃
(b) With Hot Concentrated NaOH
Chlorine reacts with hot concentrated sodium hydroxide forming sodium chloride and sodium chlorate.
3Cl₂ + 6NaOH → 5NaCl + NaClO₃ + 3H₂O
(c) With Sodium Thiosulphate (Na₂S₂O₃)
Chlorine reacts with sodium thiosulphate to produce sodium tetrathionate and sodium chloride.
Cl₂ + 2Na₂S₂O₃ → Na₂S₄O₆ + 2NaCl
03 00 AM IST - 13 Mar'26
Solved Chemistry Long Answer Question
Describe the following reactions:
(i) Acetylation
(ii) Cannizzaro Reaction
(iii) Cross Aldol Condensation
(iv) Decarboxylation(i) Acetylation
Acetylation is the process of introducing an acetyl group (CH₃CO–) into an organic compound. It is usually carried out using reagents such as acetyl chloride or acetic anhydride.
Example:
C₆H₅NH₂ + CH₃COCl → C₆H₅NHCOCH₃ + HCl(ii) Cannizzaro Reaction
The Cannizzaro reaction occurs when aldehydes without α-hydrogen atoms undergo oxidation and reduction in the presence of concentrated alkali, producing alcohol and carboxylic acid.
Example:
2HCHO + NaOH → HCOONa + CH₃OH(iii) Cross Aldol Condensation
When two different aldehydes or ketones containing α-hydrogen react in the presence of dilute base, the reaction is called cross aldol condensation.
Example:
CH₃CHO + C₆H₅CHO → C₆H₅CH=CHCHO(iv) Decarboxylation
Decarboxylation is the removal of carbon dioxide (CO₂) from a carboxylic acid or its salt.
Example:
CH₃COONa + NaOH → CH₄ + Na₂CO₃ (in presence of CaO)02 00 AM IST - 13 Mar'26
Important Solved 8-Mark Question - 2
How is ozone prepared from oxygen? Explain its reaction with:
(a) C₂H₄
(b) KI
(c) Hg
(d) PbSPreparation of Ozone
Ozone is produced by passing dry oxygen through a silent electric discharge in an ozonizer.
Reaction:
3O₂ → 2O₃Reactions of Ozone
(a) With Ethene (C₂H₄)
Ozone reacts with ethene forming an ozonide, which on hydrolysis gives aldehydes.C₂H₄ + O₃ → Ozonide → HCHO
(b) With Potassium Iodide (KI)
Ozone oxidises potassium iodide to iodine.O₃ + 2KI + H₂O → I₂ + 2KOH + O₂
(c) With Mercury (Hg)
Ozone oxidises mercury to mercuric oxide.Hg + O₃ → HgO + O₂
(d) With Lead Sulphide (PbS)
Ozone converts lead sulphide into lead sulphate.PbS + 4O₃ → PbSO₄ + 4O₂
01 00 AM IST - 13 Mar'26
Solved 8-Mark Question - 1
(a) State and explain Kohlrausch's law of independent migration of ions.
(b) What is molecularity of a reaction? How is it different from the order of a reaction? Give one example each of bimolecular and trimolecular gaseous reactions.(a) Kohlrausch’s Law of Independent Migration of Ions
Kohlrausch’s law states that at infinite dilution, each ion contributes independently to the total molar conductivity of an electrolyte. Therefore, the molar conductivity at infinite dilution is equal to the sum of ionic conductivities of the individual ions.
Example:
Λ°(NaCl) = λ°Na⁺ + λ°Cl⁻(b) Molecularity of a Reaction
Molecularity refers to the number of reacting species (atoms, ions, or molecules) that collide simultaneously in an elementary reaction step to produce a chemical change.
Difference between Molecularity and Order
• Molecularity is always a whole number and applies only to an elementary reaction step.
• Order of a reaction is obtained from the rate equation and can be zero, fractional, or a whole number.Examples
Bimolecular reaction:
H₂ + I₂ → 2HITrimolecular reaction:
2NO + O₂ → 2NO₂12 00 AM IST - 13 Mar'26
Important Long-Answer Questions for Revision
We will now begin sharing answers to some important 8-mark questions that could appear in the exam. Students can use these explanations for quick revision. Stay tuned for more solved questions.
11 00 PM IST - 12 Mar'26
Avoid These Mistakes Right Now
Since it is quite late in the evening, students should avoid starting completely new topics. Studying intensely at this hour may increase anxiety and affect concentration during the exam. Instead, try to relax and get proper sleep so you wake up fresh and ready for the TG Inter 2nd Year Chemistry exam tomorrow.
10 00 PM IST - 12 Mar'26
Important Checklist for Tomorrow’s Chemistry Exam
As the TG Inter 2nd Year Chemistry exam is scheduled for tomorrow, students should organise all the necessary items tonight to avoid any last-minute rush.
- TG Inter Hall Ticket / Admit Card
- School ID Card (if applicable)
- 2-3 Blue or Black Ball Pens
- Pencils
- Eraser
- Sharpener
- Geometry box (if required)
- Transparent water bottle
- Simple wristwatch (if permitted)
09 40 PM IST - 12 Mar'26
Another Solved 4 mark Question
Using IUPAC norms, write the formulas for the following: (a) tetrahydroxozincate(II) ion (b) hexaamminecobalt(III) sulphate (c) potassium tetrachloropalladate(II) (d) potassium tri(oxalato)chromate(III)
(a) Tetrahydroxozincate(II) ion
[Zn(OH)₄]²⁻(b) Hexaamminecobalt(III) sulphate
[Co(NH₃)₆]₂(SO₄)₃(c) Potassium tetrachloropalladate(II)
K₂[PdCl₄](d) Potassium tri(oxalato)chromate(III)
K₃[Cr(C₂O₄)₃]
09 20 PM IST - 12 Mar'26
Chlorine Manufacturing by Deacon's method
In Deacon’s process, hydrogen chloride gas is oxidised by atmospheric oxygen in the presence of a copper chloride catalyst at about 673 K to produce chlorine gas.
Chemical reaction:
4HCl + O₂ → 2Cl₂ + 2H₂O09 00 PM IST - 12 Mar'26
What is the tailing of mercury? How is it removed?
Tailing of mercury is the phenomenon in which mercury forms a thin tail while flowing on a surface due to the presence of impurities such as dust, grease, or other metals.
Removal: It can be removed by filtering mercury through a pinhole in filter paper or by treating it with dilute nitric acid, followed by washing with water.
08 40 PM IST - 12 Mar'26
What are tranquilizers?
Tranquilizers are drugs that reduce anxiety, stress, and mental tension by calming the nervous system.
Example: Diazepam or Equanil.
08 20 PM IST - 12 Mar'26
What are non-narcotic analgesics?
Non-narcotic analgesics are drugs that relieve pain without causing sleep or addiction. They are commonly used to treat mild to moderate pain.
Example: Aspirin or Paracetamol.08 00 PM IST - 12 Mar'26
What is PHBV?
PHBV (Poly-β-hydroxybutyrate-co-β-hydroxyvalerate) is a biodegradable polymer produced by microorganisms.
Uses:
- It is used for making biodegradable plastics.
- It is used in medical applications such as sutures and controlled drug release systems.
07 45 PM IST - 12 Mar'26
Roasting and Calcination
Roasting: Roasting is the process of heating sulphide ores in the presence of excess air to convert them into oxides.
Example: 2ZnS + 3O₂ → 2ZnO + 2SO₂
Calcination: Calcination is the process of heating carbonate or hydrated ores in the absence or limited supply of air to convert them into oxides.
Example: CaCO₃ → CaO + CO₂
07 30 PM IST - 12 Mar'26
Statement-Based 2 Mark Question
NH₃ forms hydrogen bonds, but PH₃ does not. Why?
Ammonia (NH₃) forms hydrogen bonds because nitrogen is highly electronegative and small in size. The N–H bond becomes strongly polar, which allows hydrogen atoms to form hydrogen bonds with the lone pair of nitrogen atoms in nearby molecules.
Phosphine (PH₃), however, does not form hydrogen bonds because phosphorus is less electronegative and larger in size than nitrogen. As a result, the P-H bond is not sufficiently polar to form hydrogen bonding.
07 15 PM IST - 12 Mar'26
One More Solved 2-Mark Question
Write equations for the carbylamine reaction of any one aliphatic amine.
Primary amines on heating with chloroform (CHCl₃) and alcoholic KOH give isocyanides. This reaction is called the carbylamine reaction.
Example (ethylamine):
C₂H₅NH₂ + CHCl₃ + 3KOH → C₂H₅NC + 3KCl + 3H₂O
The product ethyl isocyanide has a very unpleasant smell.07 00 PM IST - 12 Mar'26
Another Solved 2-Mark Question
Ammonia is a good complexing agent. Explain with an example.
Ammonia (NH₃) acts as a good complexing agent because it has a lone pair of electrons on the nitrogen atom, which it can donate to a metal ion to form a coordinate bond and produce a complex compound.
Example:
Cu²⁺ + 4NH₃ → [Cu(NH₃)₄]²⁺Here, ammonia molecules act as ligands and form a complex with the copper ion.
06 45 PM IST - 12 Mar'26
Define molarity
Molarity is defined as the number of moles of solute dissolved in one litre of solution. It is represented by M.
Formula:
M = n/V
Where
n = number of moles of solute
V = volume of solution in litres
06 30 PM IST - 12 Mar'26
Role of cryolite in the metallurgy of aluminum
Cryolite (Na₃AlF₆) is added during the extraction of aluminium in the Hall–Héroult process.
Role of cryolite:
- It lowers the melting point of alumina (Al₂O₃).
- It increases the electrical conductivity of the electrolyte, making electrolysis easier.
06 15 PM IST - 12 Mar'26
What are ambident nucleophiles?
Ambident nucleophiles are nucleophiles that have two different atoms through which they can donate an electron pair to an electrophile.
Examples:- CN⁻ can attack through carbon or nitrogen.
- NO₂⁻ can attack through nitrogen or oxygen.
06 00 PM IST - 12 Mar'26
What are enantiomers?
Enantiomers are a pair of stereoisomers that are non-superimposable mirror images of each other. They have the same molecular formula and bonding but differ in the spatial arrangement of atoms around a chiral carbon atom.
Example: The two forms of lactic acid that are mirror images of each other.
05 45 PM IST - 12 Mar'26
Amino Acid Derivatives Examples
These hormones are derived from single amino acids such as tyrosine.
Example: Adrenaline (epinephrine), Noradrenaline, Thyroxine (T₄), Triiodothyronine (T₃), Melatonin.
05 30 PM IST - 12 Mar'26
Polypeptide Hormones Examples
These hormones are made up of chains of amino acids.
Example: Insulin, Glucagon, Growth hormone, Oxytocin, Vasopressin.
05 15 PM IST - 12 Mar'26
Steroid Hormones Examples
These hormones are derived from cholesterol.
Example: Testosterone, Estrogen, Progesterone, Cortisol, Aldosterone.
05 00 PM IST - 12 Mar'26
What are hormones?
Hormones are chemical substances produced by endocrine glands and secreted directly into the bloodstream. They act as chemical messengers and regulate various physiological activities such as growth, metabolism, and reproduction.
04 45 PM IST - 12 Mar'26
Werner's Theory of Coordination Compounds
Werner’s theory explains the structure and bonding in coordination compounds. According to this theory:
- A metal atom or ion exhibits two types of valencies: primary valency and secondary valency.
- Primary valency corresponds to the oxidation state of the metal ion and is satisfied by negative ions.
- Secondary valency corresponds to the coordination number and is satisfied by ligands.
- Secondary valencies have a definite spatial arrangement around the metal ion.
Example: CoCl₃·6NH₃
According to Werner’s theory: [Co(NH₃)₆]Cl₃
In this compound:
• The primary valency of cobalt is 3, satisfied by three Cl⁻ ions.
• The secondary valency is 6, satisfied by six NH₃ molecules arranged around the cobalt ion.
04 30 PM IST - 12 Mar'26
Denaturation of Proteins
Denaturation of proteins is the process in which the natural structure of a protein is destroyed due to external factors such as heat, change in pH, chemicals, or heavy metal salts. During denaturation, the secondary and tertiary structures of the protein are disrupted, but the primary structure remains unchanged.
As a result, the protein loses its biological activity and becomes insoluble in water. Denatured proteins often coagulate or precipitate.
Examples:
• Coagulation of egg white when heated.
• Curdling of milk due to the addition of acid or during the preparation of curd.04 15 PM IST - 12 Mar'26
4-Mark Solved Numerical
A solution of glucose in water is labelled as 10% w/w. What would be the molarity of the solution?
10% w/w means 10 g of glucose is present in 100 g of solution.
Mass of glucose = 10 g
Molar mass of glucose (C₆H₁₂O₆) = 180 g mol⁻¹Moles of glucose = 10 / 180
Moles of glucose = 0.0556 molAssuming the density of the solution ≈ 1 g mL⁻¹
100 g solution ≈ 100 mL = 0.1 L
Molarity formula:
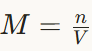
Substitute the values:
M = 0.0556 / 0.1
M = 0.556 M
Therefore, the molarity of the solution is approximately 0.56 M.
04 00 PM IST - 12 Mar'26
2-Mark Solved Numerical
Calculate the mole fraction of H₂SO₄ in a solution containing 98% w/w H₂SO₄ by mass.
Assume 100 g of solution.
Mass of H₂SO₄ = 98 g
Mass of H₂O = 2 gMolar mass of H₂SO₄ = 98 g mol⁻¹
Moles of H₂SO₄ = 98 / 98 = 1 molMolar mass of H₂O = 18 g mol⁻¹
Moles of H₂O = 2 / 18 = 0.111 molTotal moles in solution = 1 + 0.111 = 1.111 mol
Mole fraction formula:
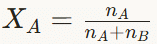
Mole fraction of H₂SO₄ = 1 / 1.111
Mole fraction of H₂SO₄ = 0.90 (approximately).
03 45 PM IST - 12 Mar'26
What is blister copper? Why is it so called?
Blister copper is the impure copper obtained during the extraction of copper after the reduction of copper oxide.
It is called blister copper because the surface of the metal shows blisters or bubbles due to the escape of sulphur dioxide (SO₂) gas during the refining process.
03 30 PM IST - 12 Mar'26
What are antiseptics?
Antiseptics are chemical substances that prevent the growth of microorganisms and are applied to living tissues such as skin and wounds to prevent infection.
Example: Dettol, iodine solution, or boric acid.
03 15 PM IST - 12 Mar'26
What is vulcanisation of rubber?
Vulcanisation is the process of heating natural rubber with sulphur at about 373–415 K to improve its strength, elasticity, and durability. Sulphur forms cross-links between rubber molecules, making the rubber harder and more elastic.
03 00 PM IST - 12 Mar'26
Faraday's first law of electrolysis
Faraday’s First Law of Electrolysis states that the amount of substance deposited or liberated at an electrode during electrolysis is directly proportional to the quantity of electricity passed through the electrolyte.
Mathematically:
m ∝ Q
or
m = ZQ
02 45 PM IST - 12 Mar'26
What is metallic corrosion?
Metallic corrosion is the slow deterioration of a metal due to chemical or electrochemical reactions with its environment such as air, moisture, or chemicals.
Example: Rusting of iron
Reaction: 4Fe + 3O₂ + xH₂O → 2Fe₂O₃·xH₂O (rust)
02 30 PM IST - 12 Mar'26
What are antacids?
Antacids are chemical substances that neutralize excess hydrochloric acid present in the stomach and give relief from acidity and indigestion.
Example: Magnesium hydroxide (Mg(OH)₂) or sodium bicarbonate (NaHCO₃).
Neutralization reaction: Mg(OH)₂ + 2HCl → MgCl₂ + 2H₂O
02 15 PM IST - 12 Mar'26
What is addition polymer?
Addition polymers are polymers formed by the repeated addition of simple unsaturated molecules (monomers) without the elimination of any small molecule.
Example reaction:
n CH₂ = CH₂ → (–CH₂–CH₂–)ₙEthene forms polyethylene, which is an addition polymer.
02 00 PM IST - 12 Mar'26
Calculate the spin-only magnetic moment of Fe²⁺ ion. (2 marks)
Electronic configuration of Fe (Z = 26):
Fe = [Ar] 3d⁶ 4s²Fe²⁺ ion loses two electrons from the 4s orbital:
Fe²⁺ = [Ar] 3d⁶Number of unpaired electrons (n) in 3d⁶ = 4
Spin only magnetic moment formula:
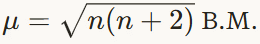
Substitute the value of n = 4
μ = √[4(4 + 2)]
Therefore, the spin only magnetic moment of Fe²⁺ ion = 4.90 Bohr Magnetons (B.M.).
μ = √(4 × 6)
μ = √24
μ = 4.90 B.M.01 45 PM IST - 12 Mar'26
4-Mark Solved Questions
Explain the terms: (i) Ligand (ii) Coordination number (iii) Coordination entity (iv) Central metal atom/ion
(i) Ligand
A ligand is an ion or molecule that donates a pair of electrons to a central metal atom or ion to form a coordinate bond in a coordination compound.
Example: NH₃, H₂O, Cl⁻, CN⁻
Example reaction: [Cu(H₂O)₆]²⁺ + 4NH₃ → [Cu(NH₃)₄]²⁺ + 6H₂O(ii) Coordination number
The coordination number is the total number of ligand donor atoms that are directly attached to the central metal atom or ion in a coordination compound.
Example: In the complex [Co(NH₃)₆]³⁺ , the coordination number of Co³⁺ is 6.(iii) Coordination entity
A coordination entity is a species that consists of a central metal atom or ion bonded to a fixed number of ligands. It behaves as a single unit in a coordination compound.
Example: [Co(NH₃)₆]³⁺(iv) Central metal atom/ion
The central metal atom or ion is the metal present at the centre of a coordination compound to which ligands are attached by coordinate bonds.
Example: In the complex [Cu(NH₃)₄]²⁺, Cu²⁺ is the central metal ion.Give the sources of the following vitamins and name the diseases caused by their deficiency: (a) A (b) D (c) E (d) K
(a) Vitamin A
Sources: Carrots, milk, butter, eggs, and green leafy vegetables.
Deficiency disease: Night blindness.(b) Vitamin D
Sources: Sunlight, fish liver oil, milk, and egg yolk.
Deficiency disease: Rickets in children and osteomalacia in adults.(c) Vitamin E
Sources: Vegetable oils, nuts, seeds, and green leafy vegetables.
Deficiency disease: Muscle weakness and reproductive disorders.(d) Vitamin K
Sources: Green leafy vegetables such as spinach and cabbage.
Deficiency disease: Poor blood clotting leading to excessive bleeding.
01 30 PM IST - 12 Mar'26
2-Mark Solved Questions
Q: What are isotonic solutions?
A: Isotonic solutions are solutions that have the same osmotic pressure at a given temperature. When two isotonic solutions are separated by a semipermeable membrane, there is no net movement of solvent between them.
Example: Blood plasma and 0.9% sodium chloride solution are isotonic.Q: Explain the terms gangue and slag.
A: Gangue: The unwanted earthy and rocky impurities, such as sand, clay, and silica, that are associated with an ore are called gangue.Slag: During the extraction of metals, flux is added to remove gangue. The flux reacts with gangue to form a fusible substance called slag, which can be easily removed from the molten metal.
01 15 PM IST - 12 Mar'26
Collision Theory LAQ
Give a detailed account of the collision theory of reaction rates of bimolecular gaseous reactions.
The collision theory explains how chemical reactions occur and why reaction rates differ. According to this theory, molecules of reactants must collide with each other for a chemical reaction to take place.
In bimolecular gaseous reactions, the reaction occurs when two molecules collide with sufficient energy and proper orientation. However, not all collisions result in a reaction. Only those collisions that have energy equal to or greater than the activation energy lead to the formation of products. These are called effective collisions.
The rate of reaction depends on the number of effective collisions between reactant molecules. The greater the number of effective collisions per unit time, the faster the reaction rate.
The rate constant k is expressed as:
k = ZAB e⁻ᴱᵃ/ᴿᵀWhere:
ZAB = collision frequency between molecules A and B
Ea = activation energy
R = gas constant
T = temperature in KelvinThus, according to collision theory, reaction rate depends on collision frequency, activation energy, and the orientation of colliding molecules. Only properly oriented collisions with sufficient energy result in the formation of products.
01 00 PM IST - 12 Mar'26
Question for Lyophilic and Lyophobic sols
What are lyophilic and lyophobic sols? Compare the two terms in terms of stability and reversibility (4 marks)
Lyophilic sols are colloidal solutions in which the dispersed phase has a strong attraction for the dispersion medium. Examples include starch and gelatin in water. These sols are highly stable and easily reversible. If the dispersion medium is removed, the sol can be formed again by simply adding the medium.
Lyophobic sols are colloidal solutions in which the dispersed phase has little or no attraction for the dispersion medium. Examples include sols of gold, sulphur, or arsenic sulphide in water. These sols are less stable and are generally irreversible. Once coagulated, they cannot easily be converted back into the sol state.
Thus, lyophilic sols are more stable and reversible, whereas lyophobic sols are less stable and irreversible.
12 45 PM IST - 12 Mar'26
Explain the extraction of zinc from zinc blende (4 marks)
Zinc is extracted from zinc blende (ZnS) by the following steps.
1. Concentration: The ore is concentrated by the froth flotation process to remove impurities.
2. Roasting: The concentrated ore is heated strongly in the presence of air to convert zinc sulphide into zinc oxide.
2ZnS + 3O₂ → 2ZnO + 2SO₂3. Reduction: The zinc oxide obtained is reduced with coke at high temperature in a furnace to produce zinc metal.
4. Refining: The crude zinc obtained is purified by distillation or electrolytic refining to obtain pure zinc.
ZnO + C → Zn + CO12 30 PM IST - 12 Mar'26
Derive Bragg's equation (4 marks)
Bragg’s equation explains the diffraction of X rays by crystals. When X rays fall on a crystal surface, they are reflected from different parallel planes of atoms in the crystal lattice.
Consider two parallel planes separated by a distance d. When a beam of X rays strikes these planes at an angle θ, part of the beam is reflected from the top plane while another part is reflected from the lower plane. The second beam travels an extra distance equal to AB + BC.
For constructive interference, the path difference must be equal to an integral multiple of the wavelength (λ).
Extra path difference = 2d sin θ
Therefore, the condition for diffraction is:
This is known as Bragg’s equation, where
nλ = 2d sin θ
n = order of reflection
λ = wavelength of X rays
d = distance between crystal planes
θ = angle of incidence.12 15 PM IST - 12 Mar'26
What are food preservatives? Give example (2 marks)
Food preservatives are chemical substances that are added to food to prevent spoilage caused by microorganisms and to increase the shelf life of food. They slow down the growth of bacteria, fungi, and other microbes.
Example: Sodium benzoate or sodium metabisulfite.
12 00 PM IST - 12 Mar'26
What is a Ziegler-Natta catalyst? (2 marks)
A Ziegler-Natta catalyst is a catalyst used in the polymerization of alkenes to produce polymers such as polyethylene and polypropylene. It is usually a combination of titanium tetrachloride (TiCl₄) and an organoaluminium compound like triethyl aluminium (Al(C₂H₅)₃). These catalysts help in forming linear and stereoregular polymers.
11 45 AM IST - 12 Mar'26
Guess Paper Set C
Students can also download Guess Paper Set C for additional practice. Solving this paper can help you revise important reactions, formulas, and theory-based questions.
11 30 AM IST - 12 Mar'26
Guess Paper Set B
Guess Paper Set B is now available for students who want to practice more expected questions before the exam. Download here!
11 15 AM IST - 12 Mar'26
Guess Paper Set A
Students preparing for the Chemistry exam can download Guess Paper Set A for quick practice.
11 00 AM IST - 12 Mar'26
TG Inter 2nd Year Chemistry Guess Papers
We will provide you with PDF links of guess papers for the chemistry exam tomorrow. These have been prepared by CollegeDekho experts after analysing the current year's expected topics and previous year questions. After that, we will start by solving important questions and providing short notes for quick revisions. Stay Tuned!
10 30 AM IST - 12 Mar'26
Model Test Paper for Chemistry
You can download the model test paper PDF for TG Inter chemistry from here!
10 15 AM IST - 12 Mar'26
How to Get "Extra Marks" & The Extra Edge
Check below some tips to get an extra advantage in your chemistry paper:
- Use a pencil to underline keywords like "isothermal," "autocatalysis," or specific catalysts like "Ziegler-Natta." It guides the examiner's eye to the "correct" parts of your answer.
- For numericals, always put your final answer (with units!) in a neat box.
- Even if not asked, draw a small, neat sketch for concepts like Schottky/Frenkel defects or the unit cell of a crystal. A labelled diagram can help you get that extra edge, even if your theoretical explanation is slightly weak.
10 00 AM IST - 12 Mar'26
Topper's Strategy to Attempt the Exam
- Use the reading time to select your 8-mark and 4-mark questions. Pick the ones where you can draw a diagram or write a clear chemical equation.
- Many toppers start with Section C (8-marks), then Section B (4-marks), and finally Section A (2-marks). This ensures you answer the high-value questions while your mind is freshest.
- Never write long, bulky paragraphs. Use bullet points for theory. For example, when explaining the "Standard Hydrogen Electrode," list: 1. Construction, 2. Working, 3. Electrode Reaction, 4. Limitations.
- In Chemistry, text is secondary; equations are primary. For every chemical property or reaction mentioned, follow it with a balanced chemical equation.
09 45 AM IST - 12 Mar'26
Chemistry High-Weightage Chapters
These chapters are expected to carry the highest weightage in the exam tomorrow:
- Electrochemistry & Chemical Kinetics - Kohlrausch’s Law, Faraday’s Laws, Nernst Equation, and differences between Order vs. Molecularity.
- P-block Elements (group 15 elements) - Haber’s Process, Contact Process, Ostwald’s Process, and Noble Gas structures
- Organic Chemistry - Named Reactions (Kolbe’s, Reimer-Tiemann, Cannizzaro, Williamson Synthesis) and Mechanism of Nucleophilic Substitution
09 30 AM IST - 12 Mar'26
What to do today?
The exam will be held tomorrow, so today is the perfect time for calm and focused revision. Instead of starting new topics, revise important formulas, named reactions, and key concepts from high-weightage chapters.
09 15 AM IST - 12 Mar'26
TG Inter 2nd Year Chemistry Syllabus - Download Here!
If you haven’t already downloaded the syllabus, you can get the complete syllabus PDF here!It’s recommended that you keep it handy during your preparations.
09 00 AM IST - 12 Mar'26
Chemistry Blueprint by Question Type
The table shows the TG Inter 2nd‑Year Chemistry 2026 blueprint, highlighting the key chapters for each question type.
Question Type
Marks per Question
Total Questions
Total Marks
Key Chapters
Long Answer (LA)
8
3
16
- Electrochemistry & Chemical Kinetics
- P-block Elements
- Organic Chemistry
Short Answer (SA)
4
8
24
- Solid State
- Solutions
- Surface Chemistry
- Metallary
- P-block Elements
- d & f block Elements
- Biomolecules
- Polymers
- Organic Chemistry
Very Short Answer (VSA)
2 marks
10
20
- Solutions
- Electrochemistry & Chemical Kinetics
- Metallary
- P-block Elements
- d & f block Elements
- Polymers
- Chemistry in Everyday Life
- Organic Chemistry












