 CUET PG Chemistry Exam 2026 March 11 LIVE
CUET PG Chemistry Exam 2026 March 11 LIVECUET PG Chemistry Exam 2026: Candidates who are in their last-minute preparation before the CUET PG Chemistry exam 2026 can refer to the short notes on the high-yield topics and some useful practice questions here. As per the schedule, the CUET PG Chemistry exam will be held from 4 PM to 5.30 PM on March 11, 2026 . Before moving to the next section, the candidates can recall the Chemistry paper pattern here. Candidates will have to answer 75 questions, which can be answered either in Hindi or in English. There will be options, and the candidates should choose the correct one. If the answer is correct, then the candidates will get +4 marks.
Note that on the CUET PG exam day, the candidates will find out the CUET PG memory-based questions and correct answers here, which will be available after concluding the exam.
CUET PG Chemistry Exam 2026: Major Highlights
Check out the major highlights related to the CUET PG Chemistry exam 2026:
Particulars | Details |
|---|---|
CUET PG Chemistry Exam | March 11, 2026 |
Exam time | 4 PM to 5.30 PM |
Medium of the exam | English and Hindi |
Total marks | 300 |
CUET PG Chemistry exam marking scheme |
|
Most weighted topics |
|
As per the experts, the test takers are advised to practice the previous years’ questions and also solve the sample question papers to have a good grip on the CUET PG Chemistry exam. For getting more updates regarding the same, stay connected with this live blog.
CUET PG Chemistry Exam 2026 March 11 LIVE
05 30 PM IST - 11 Mar'26
CUET PG Chemistry Exam 2026 Ends
CUET PG Chemistry Exam 2026 has concluded. Soon, the test takers will leave the exam hall. For getting the latest updates, the candidates are advised to stay connected with this live blog.
04 00 PM IST - 11 Mar'26
CUET PG Chemistry Exam 2026 Starts
CUET PG Chemistry 2026 exam has started. Once the exam is concluded, the exam analysis and unofficial keys along with the memory based questions will be added here.
03 00 PM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Reporting Starts
Candidates are now entering the CUET PG exam centre. Maintain the exam hall decorum and do not doany illegal work inside the exam centre.
02 00 PM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: NTA Interface Strategy
- Candidates can mark for review if they are not sure of any answer. After answering all the questions, the candidates can return to this question and review it
- Since the candidates will not be allowed to bring a scientific calculator, they can use a virtual calculator for calculations
01 00 PM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Do I bring rough sheet on my own?
No, the candidates do not need to bring any rough sheet. The same will be provided inside the exam hall to the candidates.
12 00 PM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Reporting and Entry Protocol
- Candidates should arrive at the exam centre at least 60 to 90 minutes before the gate closing time. Once the gate is closed, candidates will not be allowed to enter the exam hall.
- Do not wear large metal buckles, thick-soled shoes, or clothes
- No jewelry is allowed inside the exam hall
- Do not bring any electronic gadgets
11 00 AM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Exam Day Guidelines
- Candidates should not forget to bring their CUET PG admit card to the exam hall
- Along with the admit card, the candidates should bring their Aadhaar Card, PAN Card, Voter ID, or passport
- Carry at least two passport-sized photographs (same pasted in the application form) on the attendance sheets
- Black or blue ballpoint pen
10 00 AM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Essential Data to Memorize
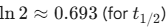
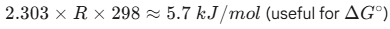
- Bragg’s Law:
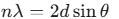
- Spin-only Magnetic Moment:
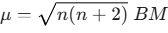
09 00 AM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Time Management Strategy
Time Segment Task Goal 0 to 30 minutes The "Scan & Strike" - Finish all the Inorganic and theoretical Organic questions here
- Do not solve heavy mathematical calculations here
30 to 80 minutes The "Numerical Grind" - Solve Physical Chemistry and mechanism-based Organic questions
80 to 90 minutes The "Review & Guess" - Review marked questions
- If you are not sure, then do not answer the questions
08 00 AM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Last Minute Preparation Tips (1/1)
Usually, the candidates make mistakes while calculating the Physical Chemistry section; hence, they should keep in mind the following instructions:
- Double-check the units in Electrochemistry and Thermodynamics.
- Quickly review the t1/2 relations for 1, 1st and 2nd order for Kinetics.
- Revise the Langmuir Adsorption Isotherm graph and the difference between Physisorption and Chemisorption.
07 00 AM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Toppers' tips to Follow (1/2)
- For answering the Organic Chemistry questions, candidates should focus more on the Mechanism over the Memory Approach.
- As per the toppers, candidates should solve the "one-liner" and theoretical questions (from Organic and Inorganic Chemistry) in the first 40 minutes
- Solve the rest Physical Chemistry Numericals in 65 minutes.
- To solve the "Match the Following" questions, the candidates should find one correct pair; usually, the 2 or 3 options will be eliminated immediately.
- It is better to avoid blind guessing, since there is a 1 mark deduction for every incorrect answer. So it's better to skip the questions.
06 00 AM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Toppers' tips to Follow (1/1)
- NTA usually mixes the units. Hence, always convert the SI units before plugging values into the Arrhenius Equation or Nernst Equation
- Don't forget the first order Kinetics, that is
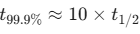 . This is the common shortcut for CUET
. This is the common shortcut for CUET - The candidates can save their crucial time during calculations if they memorize
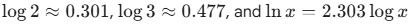
05 00 AM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Practice Questions on Chemical Bonding (1/3)
Ques. Which type of intermolecular force is primarily responsible for the high boiling point of water (H20) compared to H2S?
Answer. Hydrogen Bonding
Ques. What is the hybridization and the formal charge on the central Oxygen atom in the Ozone (O3) molecule?
Answer. sp2, +1
04 00 AM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Practice Questions on Chemical Bonding (1/2)
Ques. Using Fajan's rules, identify which of the following halides has the highest covalent character.
Answer. AlCl3
Ques. What is the hybridization of the central Phosphorus atom in PCl5 and its resulting molecular geometry?
Answer. sp3d, Trigonal bipyramidal
Ques. Which molecule possesses a non-zero dipole moment despite having polar bonds?
Answer. NH3
03 00 AM IST - 11 Mar'26
CUET PG Chemistry Exam 2026: Practice Questions on Chemical Bonding (1)
Ques. According to VSEPR theory, what is the geometry and the number of lone pairs on the central atom in the XeF4 molecule?
Answer. Square planar, 2 lone pairs
Ques. Which of the following species is paramagnetic according to Molecular Orbital Theory?
Answer. B2
02 00 AM IST - 11 Mar'26
The Periodic Trend Summary Table: Cheat Sheet for Quick Revision
Property Across a Period (----->) Down a Group 
Atomic/Ionic Radius Decreases Increases Ionization Energy Increases Decreases Electron Affinity More Negative Less Negative Electronegativity Increases Decreases Metallic Character Decreases Increases 01 00 AM IST - 11 Mar'26
Short notes on Periodicity of Elements for CUET PG Chemistry Exam 2026 (1/2)
Atomic and Ionic Radii:
- Atomic Radius decreases across the period (due to the rise in Zeff) and rises down a group (due to the new shells).
- Ionic Radius: Anions are always greater than their parent atoms (02->0). Cations are always smaller than the parent atoms (Mg2+<Mg)
- Isoelectronic Species: For the ions with a similar number of electrons, the radius decreases as the atomic number increases.
12 00 AM IST - 11 Mar'26
Short notes on Periodicity of Elements for CUET PG Chemistry Exam 2026 (1/1)
- Effective Nuclear Charge and Shielding:
- Slater's Rule: This rule is used for calculating the screening constant (
 σ); that is Zeff= Z-
σ); that is Zeff= Z- σ
σ - Trend: Zeff rises across a period (leads to the smaller atoms) and remains constant down a group.
- Slater's Rule: This rule is used for calculating the screening constant (
- Effective Nuclear Charge and Shielding:
11 00 PM IST - 10 Mar'26
Periodicity of Elements Practice questions for CUET PG Chemistry Exam 2026 (1/3)
Ques. Which of the following properties generally decreases across a period from left to right?
Answer. Atomic Radius
Ques. The electronegativity of elements on the Pauling scale is based on:
Answer. Bond dissociation energies of molecules
10 00 PM IST - 10 Mar'26
Periodicity of Elements Practice questions for CUET PG Chemistry Exam 2026 (1/2)
Ques. Why is the electron gain enthalpy of Chlorine (Cl) more negative than that of Fluorine (F)?
Answer. Chlorine has a larger atomic size.
Ques. The diagonal relationship between Beryllium (Be) and Aluminum (Al) is due to their similar:
Answer. Ionic radii and charge-to-size ratio
09 00 PM IST - 10 Mar'26
Periodicity of Elements Practice questions for CUET PG Chemistry Exam 2026 (1/1)
Ques. Which of the following elements has the highest first ionization energy?
Answer. Nitrogen.
Ques. Which factor is primarily responsible for the 'Lanthanide Contraction'?
Answer. The poor shielding effect of 4 f electrons
08 00 PM IST - 10 Mar'26
Atomic Structure Practice questions for CUET PG Chemistry Exam 2026 (1/2)
Ques. What is the total number of nodes (radial + angular) for a 4d orbital?
Answer. 3
Ques. If the velocity of a particle is doubled and its mass is halved, how does its de Broglie wavelength (λ) change?
Answer. It remains the same
07 00 PM IST - 10 Mar'26
Atomic Structure Practice questions for CUET PG Chemistry Exam 2026 (1/1)
Ques. For a hydrogen atom, which of the following expressions correctly represents the energy of an electron in the $n$-th orbit according to the Bohr model?
Answer. En= -13.6/n2 eV
Ques. The number of radial nodes in a 3p orbital is:
Answer: 1
06 00 PM IST - 10 Mar'26
CUET PG Chemistry Exam 2026: Common Trap to Avoid in Electrochemistry Section
The "Half-Cell" Trap:
Noone should add Eo values for finding out the potential of a third half-reaction. Candidates must add the ΔG0 values.
- Correct: ΔG03= ΔG01+ΔG02
- Incorrect: E03= E01+E02 (unless n is the same for all)
05 00 PM IST - 10 Mar'26
3 One-Liners for Kinetics
- Half-Life Ratio: For a first-order reaction, the time taken for 99.9% completion will be approximately 10 times the half-life (t99.9%
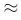 10Xt1/2)
10Xt1/2) - Units of k: If the units of k are L2mol-2s-1, the reaction is third order
- Activation Energy: If Ea=0, the rate constant k becomes equivalent to the pre-exponential factor A and is independent of temperature.
- Half-Life Ratio: For a first-order reaction, the time taken for 99.9% completion will be approximately 10 times the half-life (t99.9%
04 00 PM IST - 10 Mar'26
Numerical problem on The Reversible Intermediate (Lindemann Mechanism)
This is a classic model for unimolecular reactions:
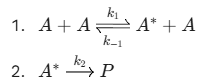
Question: Derive the rate law for the formation of product P, assuming A* is at steady state.
03 00 PM IST - 10 Mar'26
Numerical problem on "Steady State Approximation"
Problem 1: The Multi-Step Mechanism
Consider the following decomposition of a reactant A
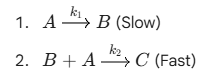
Question: If B is a highly reactive intermediate, what is the rate of formation of $C$ using the Steady State Approximation?
Conclusion: The reaction is First Order with respect to A.
02 00 PM IST - 10 Mar'26
CUET PG Chemistry Exam 2026: Exam Strategy for Chemical Kinetics
- Unit Watch: The units of k change with order.
- Zero order: mol L-1 s-1
- First order: s-1
- Second order: L mol-1s-1
- Graph Identification: A plot of log k vs 1/T is a straight line with slope - Ea/2.303 R
- Unit Watch: The units of k change with order.
01 00 PM IST - 10 Mar'26
Pro Guidance Tips on Chemical Kinetics: Rate Laws and Order of Reaction
Zero Order: Rate is independent of concentration:
- Integrated Rate Law: [A]= [A]0-kt
- Half-life: [A]0/2k (Directly proportional to initial concentration).
First-Order: Rate is proportional to concentration.
- Integrated Rate Law:
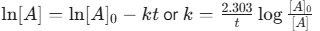
- Half-life: 0.693/k (Independent of initial concentration)
n-th Order Half-life:
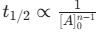 (Crucial for identifying order from data).
(Crucial for identifying order from data).12 00 PM IST - 10 Mar'26
"One-Liner" questions on Phase Equilibria (Clausius-Clapeyron equation) (1/2)
Ques. For a two-component system at constant pressure, what is the 'Reduced' or 'Condensed' Phase Rule? Answer. F=C-P+1
Ques. For a one-component system, what is the maximum number of phases that can coexist at equilibrium? Answer. 3
Ques. Which assumption is made when deriving the Clausius-Clapeyron equation from the Clapeyron equation? Answer. The vapor behaves as an ideal gas, and its molar volume is much larger than the liquid's
Ques. For the sublimation of iodine, which equation would you use to find the temperature dependence of its vapor pressure? Answer. Clausius- Clapeyron equation with ΔHsub
12 00 PM IST - 10 Mar'26
Numerical Problem on Thermodynamic Potentials & Maxwell Relations
Ques. Which of the following is equivalent to (δG/ δP)T?
- -S
- V
- T
- -P
Answer. Option 2
11 00 AM IST - 10 Mar'26
"One-Liner" questions on Phase Equilibria (Clausius-Clapeyron equation) (1/1)
- According to the Gibbs Phase Rule (F= C-P+2), what are the degrees of freedom for a system of pure water at its triple point? Ans. 0
- Which form of the Clausius-Clapeyron equation correctly relates the vapor pressure (P) to temperature (T)? Ans.
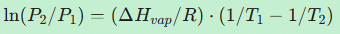
- For the water system, why is the slope of the solid-liquid coexistence line (dP/dT) negative? Ans. The molar volume of ice is greater than that of liquid water
- At the critical point of a substance, which phenomenon is observed? Ans. The distinction between liquid and gas disappears
10 00 AM IST - 10 Mar'26
Numerical Problem on Gibbs-Helmholtz and Spontaneity
Problem Type: For a certain reaction, ΔH= -40kJmol-1 and ΔS= -100 JK-1 mol-1. At what temperature does the reaction become non-spontaneous?
Expert Solution:
- Identify the Equilibrium Point: A reaction changes spontaneity at ΔG=0
- Set up the equation:
- 0=ΔH - TΔS------> T= ΔH/ΔS
- Check Units (The Trap): ΔH is in kJ, ΔS is in J. Convert ΔH to J. T= (-40000J)/(-100 JK-1)= 400 K
- Analyze the Trend: Since both ΔH and ΔS are negative, the reaction is spontaneous at low temperatures (T<400 K) and becomes non-spontaneous at T>400 K.
09 00 AM IST - 10 Mar'26
Numerical problems on Carnot Efficiency
Problem Type: A Carnot engine operates between a source at 527 °C and a sink at 127 °C. If it absorbs 1000 J of heat from the source, how much work is done?
Expert Solution:
- Convert to Kelvin (Mandatory):
- Thot= 527+273= 800 K
- Tcold= 127+273= 400 K
- Calculate Efficiency= 1- Tcold/Thot= 1- (400/800)= 0.5 (or 50%)
- Calculation of Work (W):
- Efficiency= W/qin------> 0.5= W/1000
- W= 500J
- Convert to Kelvin (Mandatory):
08 45 AM IST - 10 Mar'26
CUET PG Chemistry Exam 2026: Expert Tips for Thermodynamics
- State vs. Path Functions: Candidates should note it down that q and w are path functions; whereas U, H, S, G, and A are state functions.
- Cyclic Process: For the cyclic process, the change in any state function is always zero.
- Efficiency of Carnot Engine: n=1-Tcold/Thot=w/qhot. Temperatures must always be in Kelvin.
08 30 AM IST - 10 Mar'26
CUET PG Chemistry Ionic Equilibria: Final Revision Checklist
- Log Values: Memorize log 2(0.3), log 3 (0.48), and log 5 (0.7)
- Alpha (α): Candidates should check if the approximation 1-α
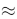 1 is valid (usually if Ka/C<10-3)
1 is valid (usually if Ka/C<10-3) - Temperature Dependency: Keep in mind that Kw rises when the temperature rises, which means pH+pOH will be less than 14 at temperatures above 298 K.
08 15 AM IST - 10 Mar'26
CUET PG Chemistry Ionic Equilibria: Indicators and Titration
Particulars Details Strong Acid vs. Strong Base Any indicator (Phenolphthalein or Methyl Orange) Weak Acid vs. Strong Base Phenolphthalein (Equivalence point pH>7) Strong Acid vs. Weak Base Methyl Orange (Equivalence point pH<7) 08 00 AM IST - 10 Mar'26
Salt Hydrolysis: The Shortcut Table
Salt Type Example Nature SA+SB NaCl Neutral WA+SB CH3COONa Basic SA+WB NH4Cl Acidic WA+WB CH3COONH4 Variable 07 45 AM IST - 10 Mar'26
CUET PG Chemistry Ionic Equilibria: The "Dilution Trap" in pH Calculations
- The Concept: The candidates cannot simply take -log(10-8). They should account for the [H+] contributed by the auto-ionization of water (10-7M)
- The Math: Total [H+]= [H+]acid+ [H+]water
- The Result: The pH of 10-8 M HCl is approximately 6.98, not 8. An acidic solution would not be a basic pH.
07 30 AM IST - 10 Mar'26
Quick list of "One-Liner" facts for Organometallic Chemistry (Inorganic) (1/4)
18-Electron Calculation Tip: For the Neutral Atom Method, are highlighted in the following section-
CO= 2 electrons
n5-Cp= 5 electrons
CH3= 1 electron
PPh3= 2 electrons
07 15 AM IST - 10 Mar'26
Quick list of "One-Liner" facts for Organometallic Chemistry (Inorganic) (1/3)
- Reductive Elimination: This is the exact opposite of the oxidative addition, and this is the final step of many cycles (such as Monsanto Process), where the product is released.
- Oxidative Addition: During this step of the catalytic cycle, both the formal oxidation state and the coordination number of the metal will be increased by two.
- Monsanto vs. Cativa Process: Both produce the Acetic Acid; nonetheless, Monsanto uses a Rhodium catalyst, while the Cativa process uses a more efficient Iridium catalyst.
07 00 AM IST - 10 Mar'26
Quick list of "One-Liner" facts for Organometallic Chemistry (Inorganic) (1/2)
- IR Stretching Frequency (vCO): As π-backbonding rises, the M-C bond becomes stronger, but the C
 O bond becomes weaker, and decreases the IR stretching frequency.
O bond becomes weaker, and decreases the IR stretching frequency. - Wilkinson’s Catalyst: It is a 16-electron, square planar complex used for the hydrogenation of alkenes. The formula is represented as
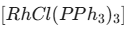
- Zeise’s Salt: It was the first organometallic compound to involve an alkene ligand, and the formula is
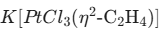
- IR Stretching Frequency (vCO): As π-backbonding rises, the M-C bond becomes stronger, but the C
06 45 AM IST - 10 Mar'26
Quick list of "One-Liner" facts for Organometallic Chemistry (Inorganic) (1/1)
- The 18-Electron Rule: The most stable transition metal organometallic complexes, such as Metal Carbonyls, follow the 18-Electron rule. This is equivalent to the octet rule.
- Synergic Bonding: In Metal Carbonyls, (M-CO), strength is gained by CO----->m σ- donation and m----->CO λ-backbonding.
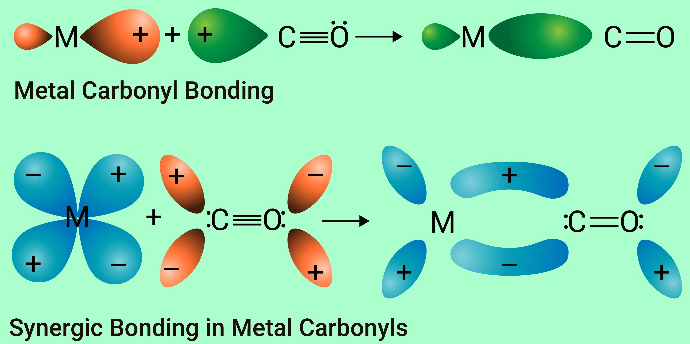
06 30 AM IST - 10 Mar'26
CUET PG Chemistry Exam 2026: Emergency Revision Formula (1/3)
- Particle in a 1D Box:
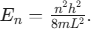 Note that energy is proportional to n2 and inversely proportional to L2.
Note that energy is proportional to n2 and inversely proportional to L2. - Bragg’s Law: nλ= 2dsinθ
- Colligative Properties:
- Boiling point elevation: ΔTb=iKbm
- Freezing point depression: ΔTf=iKfm
- Particle in a 1D Box:
06 15 AM IST - 10 Mar'26
CUET PG Chemistry Exam 2026: Emergency Revision Formula (1/2)
Chemical Kinetics & Electrochemistry:
- Zero Order:
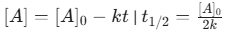
- First Order:
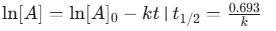
- Arrhenius Equation:
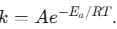 . A common question involves calculating Ea from two different temperatures.
. A common question involves calculating Ea from two different temperatures. - Nernst Equation:
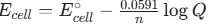 (at 298 K)
(at 298 K) - Kohlrausch’s Law:
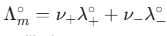 . Very useful for calculating the molar conductivity of weak electrolytes at infinite dilution.
. Very useful for calculating the molar conductivity of weak electrolytes at infinite dilution.
- Zero Order:
06 00 AM IST - 10 Mar'26
CUET PG Chemistry Exam 2026: Emergency Revision Formula (1/1)
Thermodynamics & Chemical Equilibrium:
- First Law: ΔU= q+w (Remember: w= -PΔV for expansion)
- Entropy (ΔS): Know the formula for phase changes, ΔS= qrev/T, and for ideal gas expansion, ΔS= nRIn(V2/V1)+nCvIn (T2/T1)
- Gibbs Free Energy: ΔG= ΔH-TΔS [If ΔG<0, the reaction is spontaneous]
- Van't Hoff Equation: In(K2/K1)= ΔH0/R (1/T1-1/T2). Use this for temperature dependence of equilibrium constants.
05 45 AM IST - 10 Mar'26
CUET PG Chemistry Exam 2026: Constants to Remember
For Energy/Work: R= 8.314 JK-1mol-1
For Gas Laws/ Pressure: R= 0.0821 atm K-1mol-1
Faraday’s Constant: F= 96500 C mol-1
05 30 AM IST - 10 Mar'26
CUET PG Chemistry Exam 2026: Expert Tip for Final Revision
As per the recent paper pattern, the general section that is Part A was removed in the recent cycle, and the competition will be done solely on the Chemistry Knowledge.
- Candidates should be careful with the units in Physical Chemistry. A small mistake in converting R (Gas Constant) values will lead to choosing the incorrect option.
- Solve previous years' papers: It is wise to solve the 2025 and 2024 question papers, since NTA sometrimes repeat the conceptual patterns.
05 15 AM IST - 10 Mar'26
What are the high yields Organic Chemistry Topics? (1/2)
Chapter Names High-Yields Topics Organic Spectroscopy Structure elucidation using NMR (1H and 13C), IR and UV-Vis Natural Products & Biomolecules SN1, SN2, E1, E2, and Electrophilic Aromatic Substitution (EAS) Functional Groups Amino acids, peptides, proteins, and carbohydrates 05 00 AM IST - 10 Mar'26
What are the high yields Organic Chemistry Topics? (1/1)
Chapter Names High-Yields Topics Stereochemistry Chirality, R/S and E/Z nomenclature, and conformational analysis of cyclohexane (chair/boat forms) Reaction Mechanisms SN1, SN2, E1, E2, and Electrophilic Aromatic Substitution (EAS) Named Reactions Hemoglobin, myoglobin, and the Na/K pump 04 45 AM IST - 10 Mar'26
What are the high yields Inorganic Chemistry Topics? (1/2)
Chapter Names High-Yields Topics Transition (d-block) & Inner Transition (f-block) Elements Lanthanide contraction, magnetic properties, and color spectra Organometallic Compounds 18-electron rule, metal carbonyls, and hapticity Bioinorganic Chemistry Hemoglobin, myoglobin, and the Na/K pump 04 30 AM IST - 10 Mar'26
What are the high yields Inorganic Chemistry Topics? (1/1)
Chapter Names High-Yields Topics Coordination Chemistry Crystal Field Theory (CFT), splitting in octahedral/tetrahedral complexes, CFSE, Jahn-Teller distortion, and isomerism Chemical Bonding VSEPR theory, hybridization, and Molecular Orbital (MO) diagrams (especially for CO, NO, and O2). Main Group Elements (s and p block) Borazines, silicates, phosphazenes, and noble gas compounds 04 15 AM IST - 10 Mar'26
What are the high yields Physical Chemistry Topics? (1/2)
Chapter Names High-Yields Topics Electrochemistry Nernst equation, Kohlrausch law, Debye-Hückel theory, and electrochemical cells Atomic Structure Bohr’s model, quantum numbers, and radial/angular distribution curves Surface Chemistry & Phase Equilibria Adsorption isotherms (Langmuir, BET) and Gibbs phase rule 04 00 AM IST - 10 Mar'26
What are the high yields Physical Chemistry Topics? (1/1)
The Physical Chemistry section contains 30% to 35% weightage of the paper, and this is the numerical and conceptual section. Check out the chapter-wise most important topics here:
Chapter Names High-Yields Topics Quantum Chemistry Schrödinger equation, operators, particle in a box, hydrogen atom, and wave functions Thermodynamics Laws of thermodynamics, Gibbs free energy, entropy, Maxwell relations, and spontaneity Chemical Kinetics Rate laws, collision theory, Arrhenius equation, and steady-state approximation












